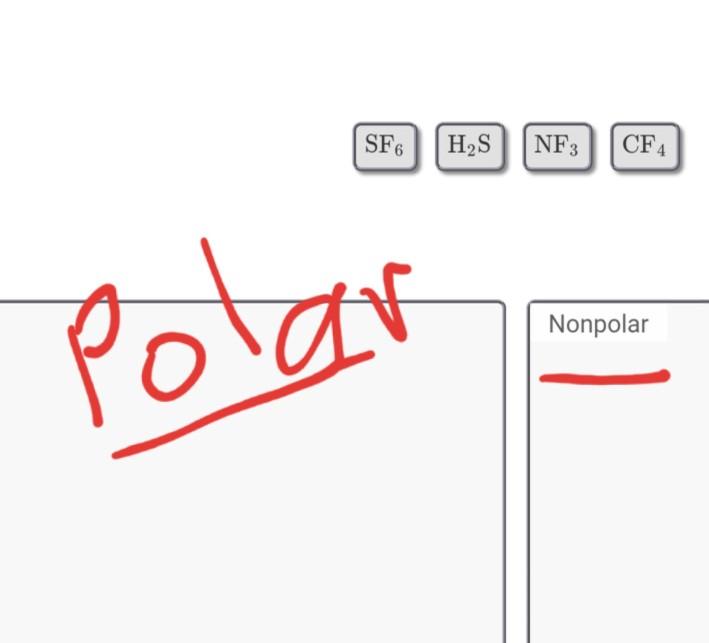
![SOLVED: Determine whether each mol ecule given below is polar or nonpolar; Drag the appropriate items to their respective bins: Resed] Help Polar Nonpolar CF4 NF3 OFz SF6 SOLVED: Determine whether each mol ecule given below is polar or nonpolar; Drag the appropriate items to their respective bins: Resed] Help Polar Nonpolar CF4 NF3 OFz SF6](https://cdn.numerade.com/ask_images/e626b2904f984dbf8e0fadc01fdc9bbb.jpg)
SOLVED: Determine whether each mol ecule given below is polar or nonpolar; Drag the appropriate items to their respective bins: Resed] Help Polar Nonpolar CF4 NF3 OFz SF6
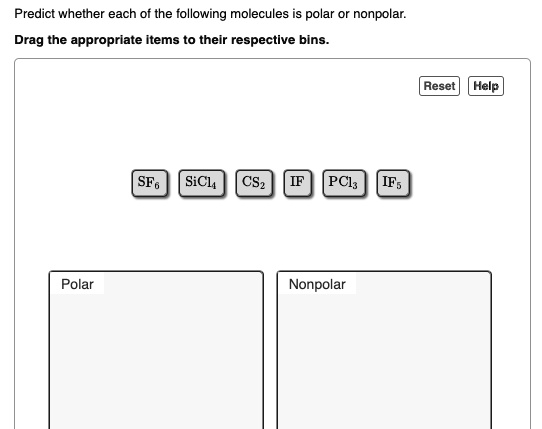
SOLVED: Predict whether each of the following molecules is polar or nonpolar: Drag the appropriate Items to their respective bins: Aeset Help SF6 SiCl CS2 PClz Polar Nonpolar IF6

Is CF4 Polar or Non-polar? (Carbon Tetrafluoride) | Is CF4 Polar or Non-polar? (Carbon Tetrafluoride) CF4 is a potent greenhouse gas and the compound is used as a low refrigerant. To determine

Which of the following molecules could exist in both polar and nonpolar forms, depending on the arrangement of the halogen atoms? a. XeFCl b. XeCl2F2 c. SiCl2F2 | Homework.Study.com

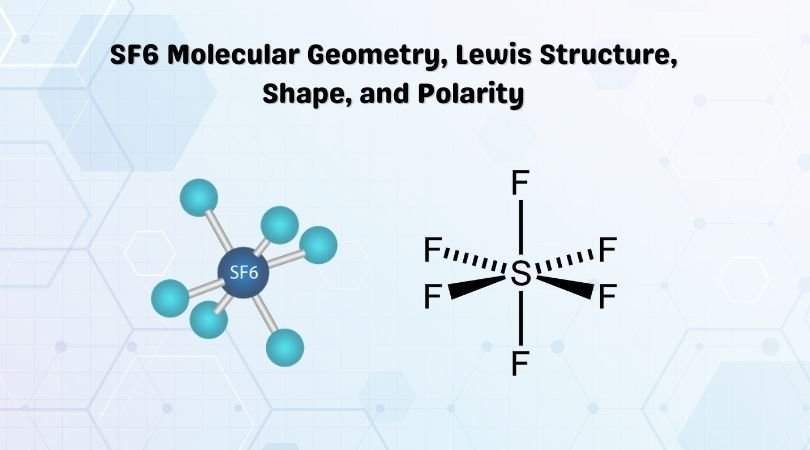
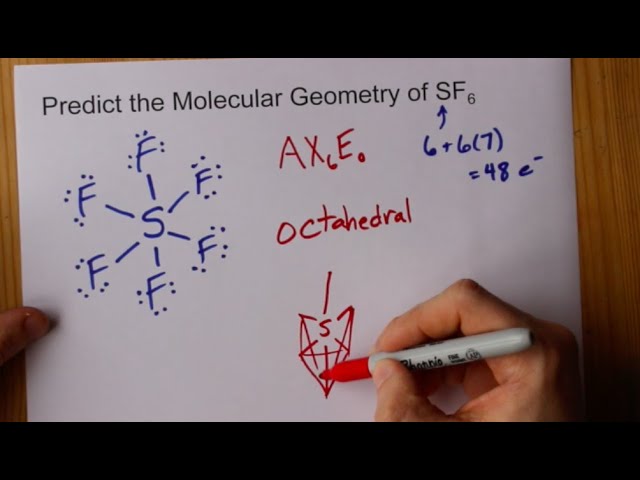
SOLVED: the electron geometry, the molecular geometry, and the polarity: SF6 Number of valence electrons: Electron geometry: Octahedral Molecular geometry: Octahedral Polarity: nonpolar For geometries, use the table in Appendix F This
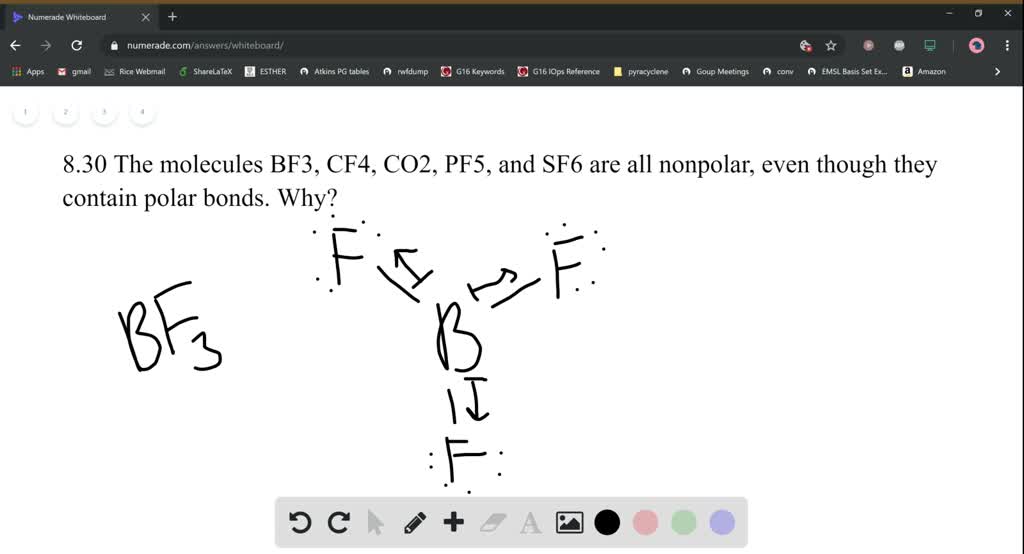
SOLVED:The molecules BF3, CF4, CO2, PF5, and SF6 are all nonpolar, even though they contain polar bonds. Why?