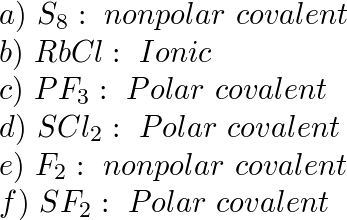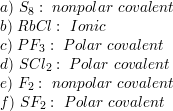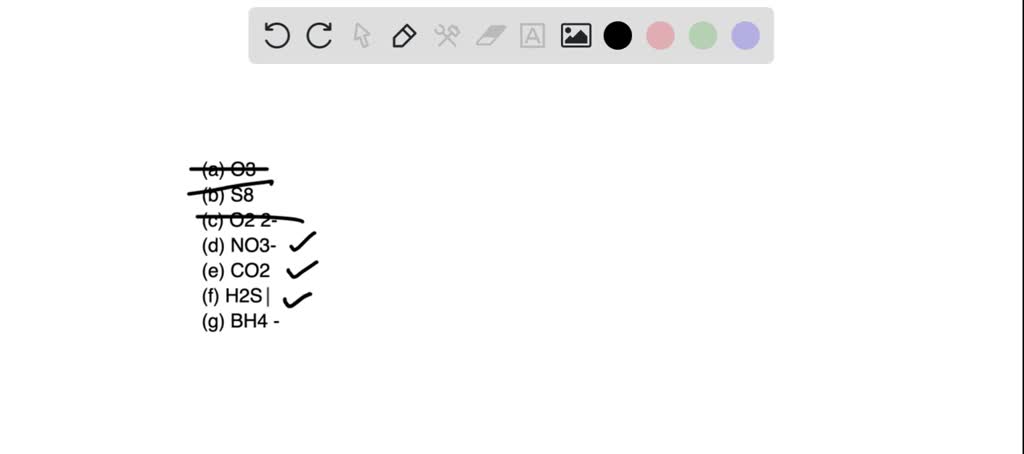
SOLVED: 22. Which of the following molecules or ions contain polar bonds? (a) O3 (b) S8 (c) O2 2− (d) NO3 − (e) CO2 (f) H2S (g) BH4 − My answers are:

Fig. S8.1: χ (2) eff across the Li K-edge comparison between systems... | Download Scientific Diagram

22 | Which of the following molecules or ions contain polar bonds? O3, S8, O2 2-, NO3-, CO2, H2S - YouTube

Mechanism and energetics of O and O2 adsorption on polar and non-polar ZnO surfaces: The Journal of Chemical Physics: Vol 144, No 18

Polar–Nonpolar Transition-Type Negative Thermal Expansion with 11.1% Volume Shrinkage by Design | Chemistry of Materials

Measurement and Correlation of the Solubilities of Sulfur S8 in 10 Solvents | Journal of Chemical & Engineering Data

Facile Access to Polar-Functionalized Ultrahigh Molecular Weight Polyethylene at Ambient Conditions | CCS Chem
Why does sulfur form S8 and not S6? Isn't the six-membered ring supposed to be more stable than the eight-membered ring. - Quora

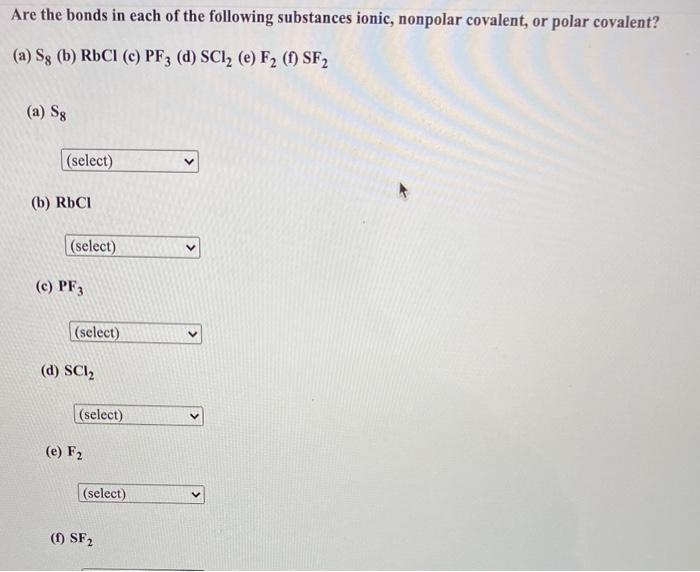
SOLVED:Are the bonds in each of the following substances ionic, nonpolar covalent, or polar covalent? Arrange the substances with polar covalent bonds in order of increasing bond polarity: (a) S8 (b) RbCl (

Amino Acid Specific Nonenzymatic Montmorillonite‐Promoted RNA Polymerization - Namani - 2021 - ChemSystemsChem - Wiley Online Library
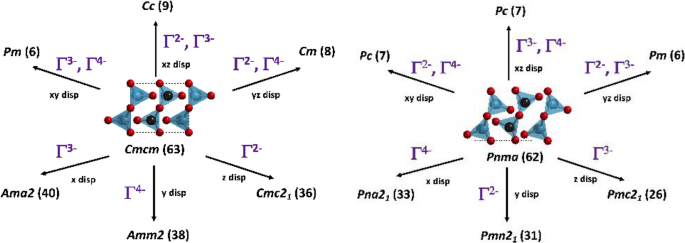
Developing new antiferroelectric and ferroelectric oxides and chalcogenides within the A2BX3 family | SpringerLink