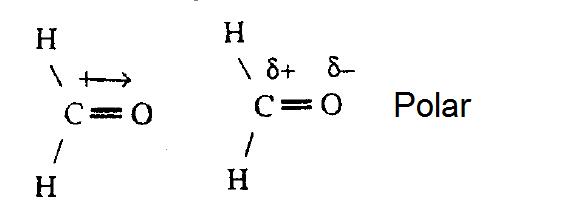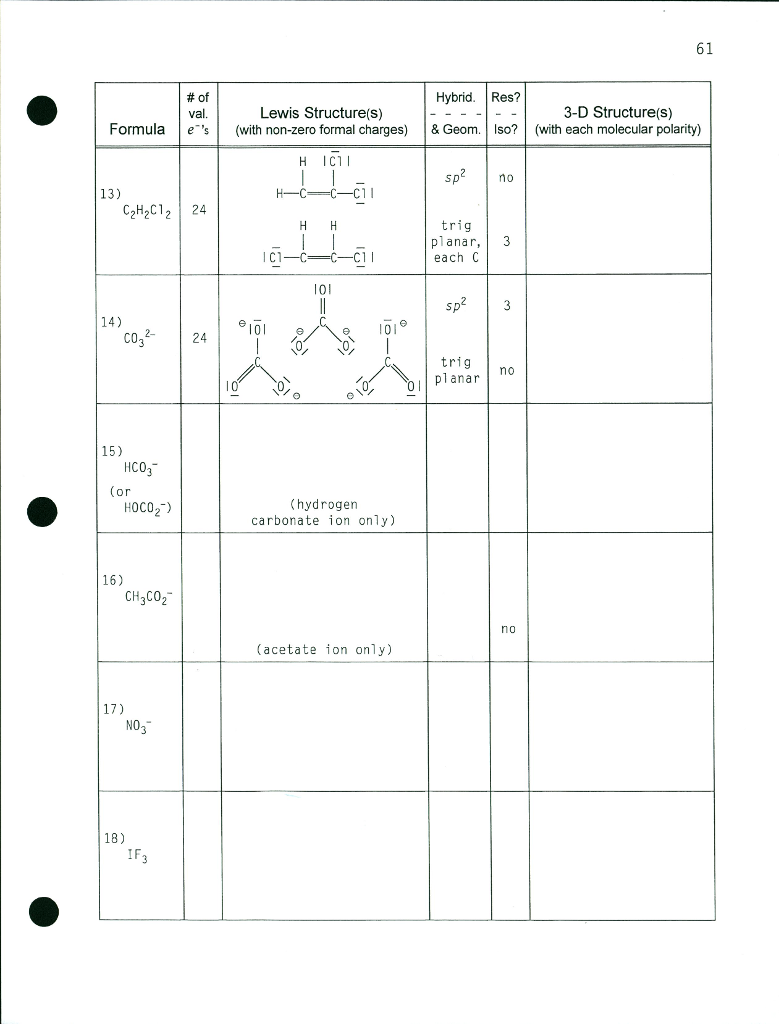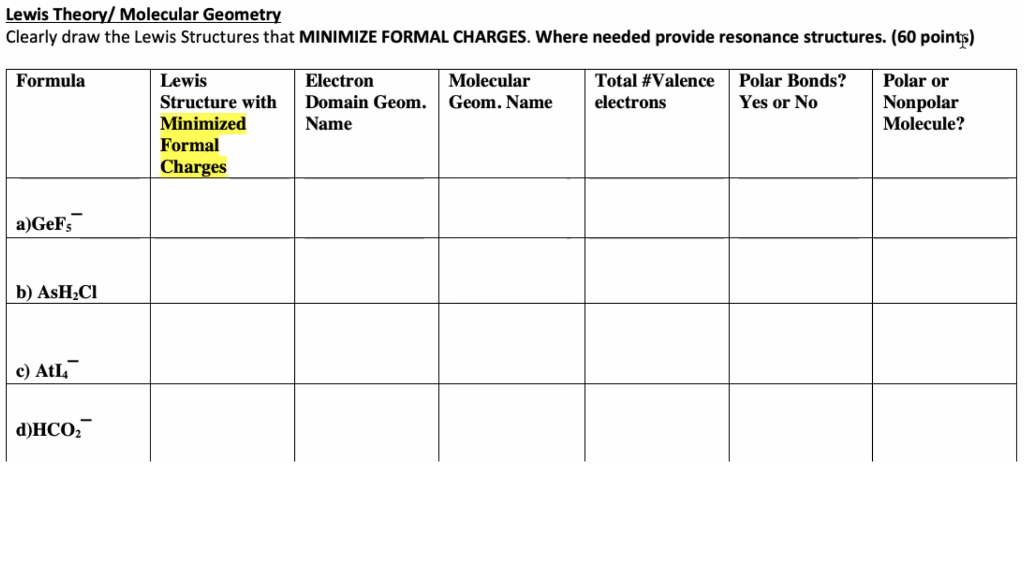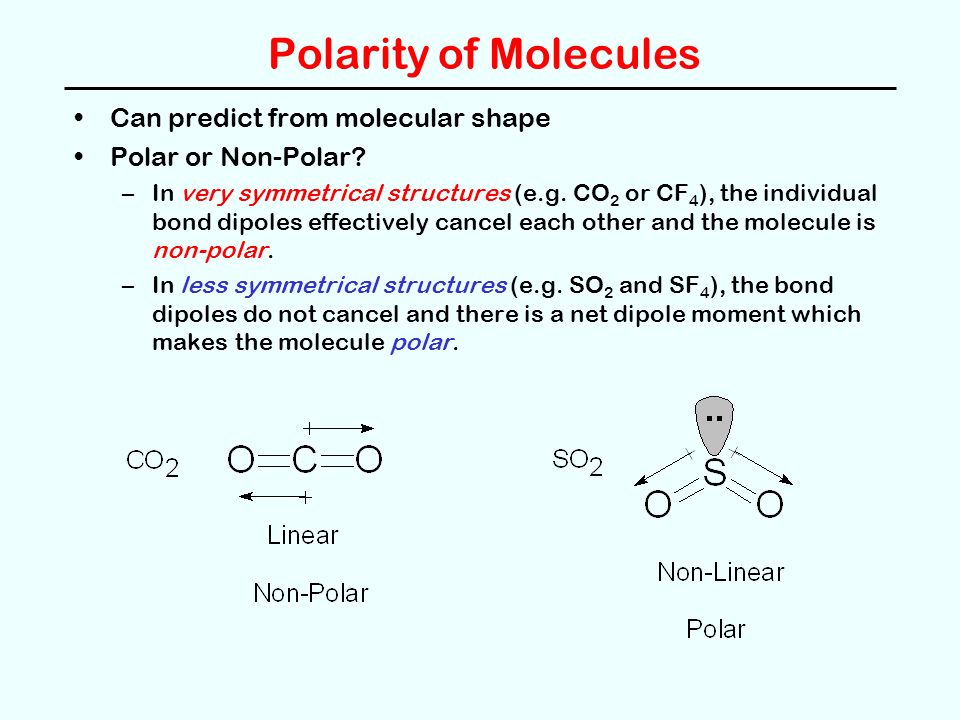
Chemical Bonding and Molecular Structure (Ch. 10) Molecular Structure General Summary -- Structure and Bonding Concepts octet rule VSEPR Theory Electronegativity. - ppt download
Molecular Geometry Examples and Questions Multiple Bonds Larger Covalent Molecules Identify procane (an aesthetic)
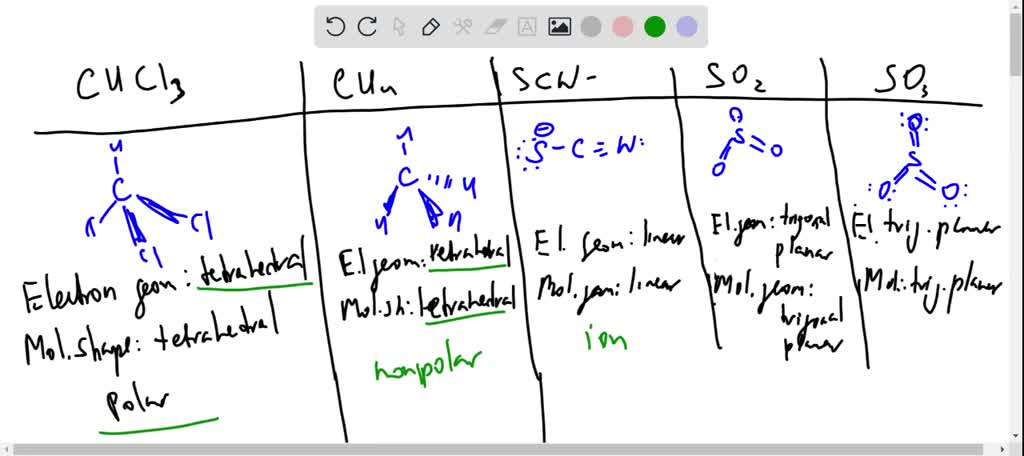
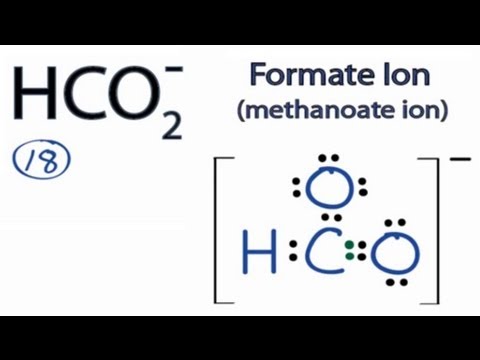
SOLVED: CO32- Electron Geometry Molecular Shape polar, non-polar, an ion 8.H2O Electron Geometry Molecular Shape polar, non-polar, an ion 9.HCO2 - Electron Geometry Molecular Shape polar, non-polar, an ion 10. H2CCHF Electron

Draw the Lewis structure for HCO2- and determine the formal charge of each atom. | Homework.Study.com