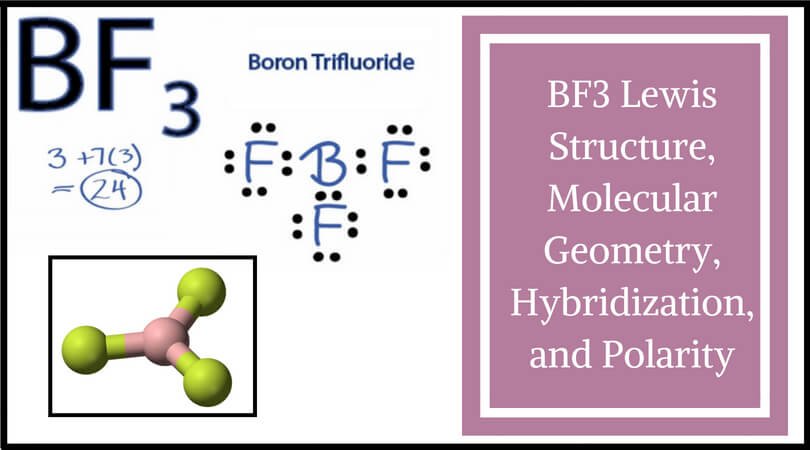
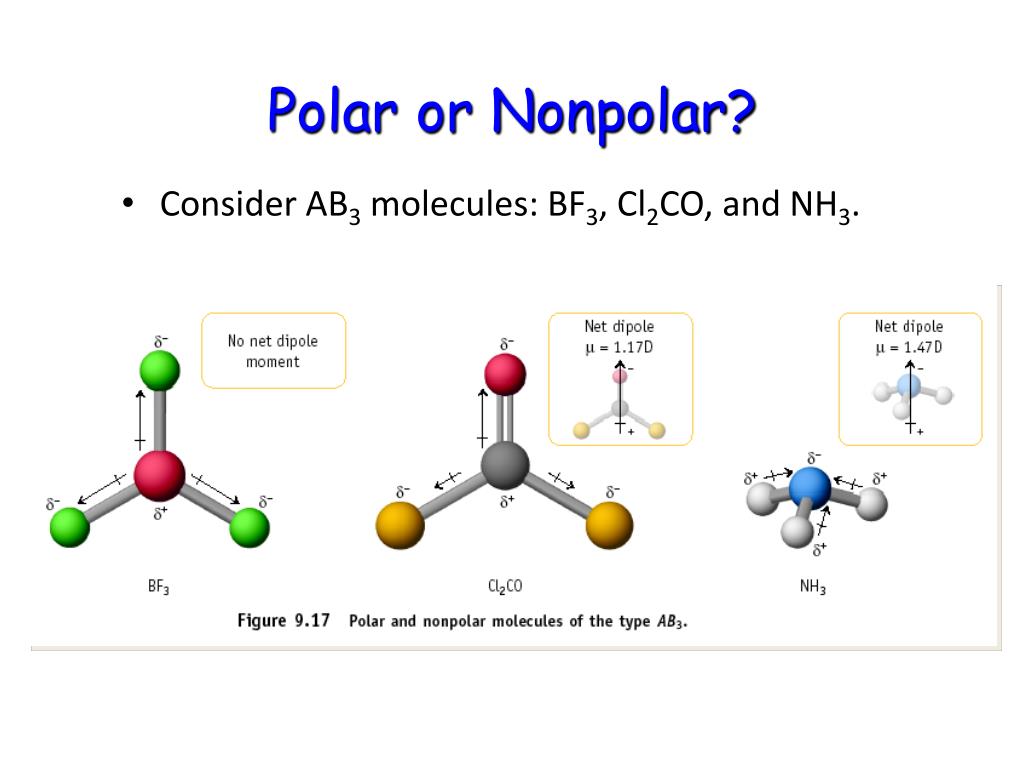
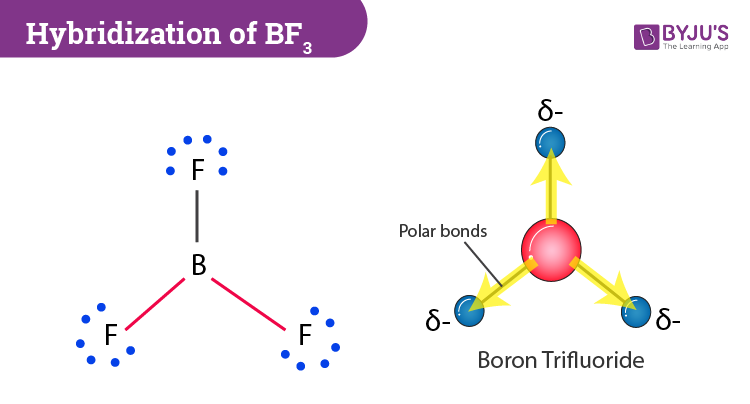
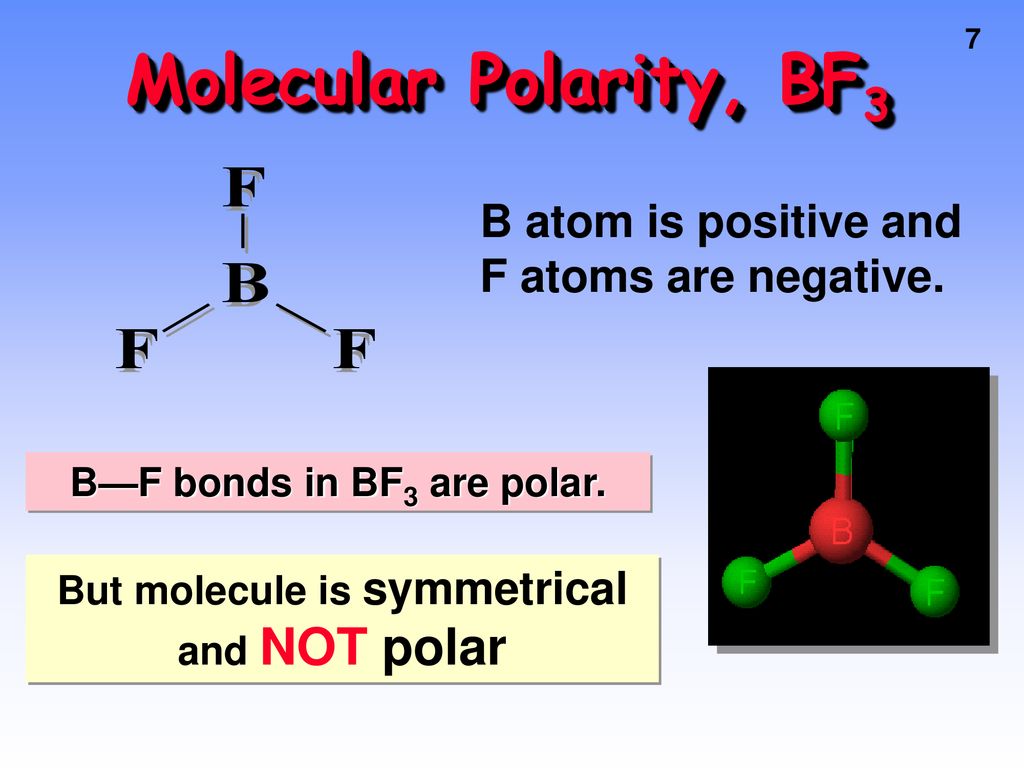
Boron Trifluoride BF3 is a non polar molecule whereas ammonia NH3is a polar molecule. The difference in polarity is related to the fact that

BF3 Molecular Geometry, Shape and Bond Angles (Boron Trifluoride) | Molecular geometry, Molecular, Molecules

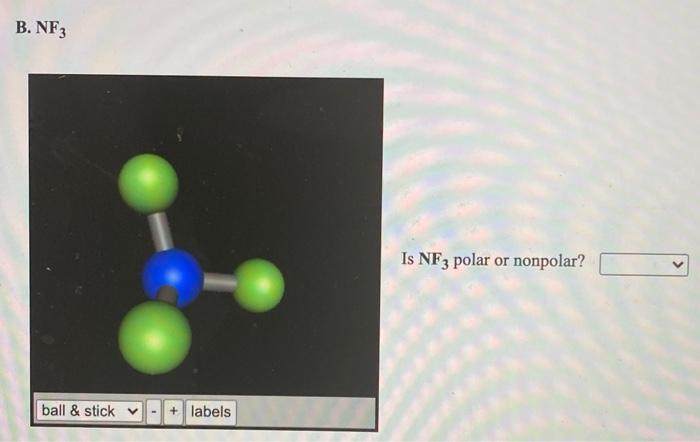
BF3 and NF3 both are covalent compounds but NF3 is polar whereas BF3 is non - polar. This is because :